Hey there, future scientists! If you’ve ever looked at the Periodic Table and felt like you were staring at a cryptic puzzle, don’t worry—you’re not alone. Starting Class 10 Chemistry can feel like learning a new language, but once you understand how atoms are structured, everything clicks into place. Today, we’re breaking down the essentials: Atomic Number, Energy Shells, and the trickiest part of all—Valency.
Understanding these isn’t just about passing your boards; it’s about understanding how the very matter around us sticks together. Let’s break it down.
1. The Atomic Number (Z)
Think of the Atomic Number as an atom’s unique ID card. No two different elements have the same atomic number.
- Definition: The Atomic Number is the total number of protons found in the nucleus of an atom.
- Symbol: It is denoted by the letter Z.
- The Golden Rule: In a neutral atom, the number of protons is equal to the number of electrons. So, if you know Z, you know how many electrons are buzzing around that nucleus!
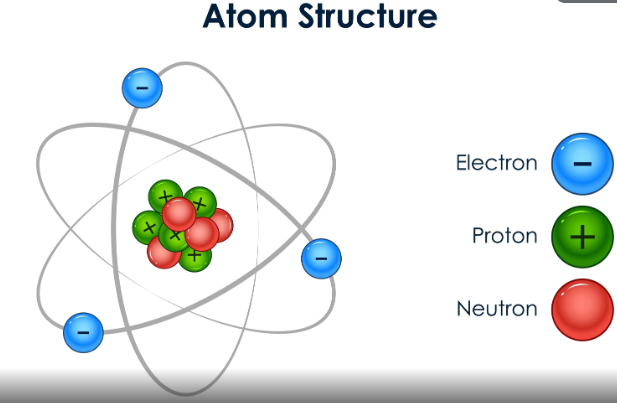
Example: Carbon has 6 protons in its nucleus. Therefore, its Atomic Number (Z) is 6.
2. The Bohr Model: Energy Levels (Shells)
Electrons don’t just swarm randomly; they live in specific “neighborhoods” called shells or energy levels. These are labeled as K, L, M, and N.
Each shell has a maximum capacity calculated by the formula 2n2, where n is the orbit number:
- K Shell (n=1): Max 2 electrons
- L Shell (n=2): Max 8 electrons
- M Shell (n=3): Max 18 electrons (though it stabilizes with 8 in early elements)
3. Valency: The “Combining Capacity”
If the atomic number is the ID card, Valency is the “social personality” of the atom. It determines how many “hands” an atom has to hold onto other atoms.
- Definition: Valency is the combining capacity of an element. It is determined by the number of valence electrons (the electrons in the outermost shell).
- The Octet Rule: Most atoms want a full outer shell (usually 8 electrons) to become stable. They will gain, lose, or share electrons to reach this “happy” state.
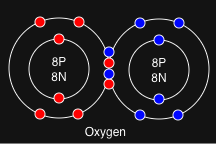
Real-World Examples
Let’s look at two classic examples you’ll definitely see in your CBSE exams:
| Element | Symbol | Atomic Number (Z) | Electronic Configuration (K,L,M) | Valence Electrons | Valency |
|---|---|---|---|---|---|
| Magnesium | Mg | 12 | 2, 8, 2 | 2 | 2 (Loses 2) |
| Chlorine | Cl | 17 | 2, 8, 7 | 7 | 1 (8-7=1) |
Why does this matter?
When Magnesium (Mg) meets Chlorine (Cl), Magnesium wants to get rid of 2 electrons, and each Chlorine atom wants to take 1. This is why they form MgCl_2! By knowing the valency, you can predict the formula of almost any chemical compound.
4. Special Cases: Hydrogen and Helium
These two are the “exceptions” because they only deal with the K shell.
Hydrogen (H)
- Atomic Number: 1
- Configuration: K = 1
- Valency: 1. It needs one more electron to fill its K shell (duplet).
Helium (He)
- Atomic Number: 2
- Configuration: K = 2
- Valency: 0. Since the K shell can only hold 2 electrons, Helium is already “full” and stable. It has no desire to react, making it a Noble Gas.
5. Quick Examples for Practice
| Element | Atomic Number | Electronic Configuration | Valence Electrons | Valency |
|---|---|---|---|---|
| Lithium (Li) | 3 | 2, 1 | 1 | 1 |
| Oxygen (O) | 8 | 2, 6 | 6 | 2 (8 – 6) |
| Neon (Ne) | 10 | 2, 8 | 8 | 0 |
Test Your Knowledge (MCQs)
Q1. An element has an atomic number of 13. What is its valency? A) 1 B) 2 C) 3 D) 5
Q2. Why is the valency of Helium zero, even though it has 2 electrons? A) It is a metal. B) Its K shell is completely filled (Duplet). C) It has no protons. D) It belongs to the 2nd period.
Q3. Which of the following elements has a valency of 2? A) Sodium (Atomic No. 11) B) Magnesium (Atomic No. 12) C) Aluminum (Atomic No. 13) D) Carbon (Atomic No. 6)
Q4. If an atom has 7 electrons in its outermost shell, its valency will be: A) 7 B) 8 C) 1 D) 0
Provide your answers in comments

